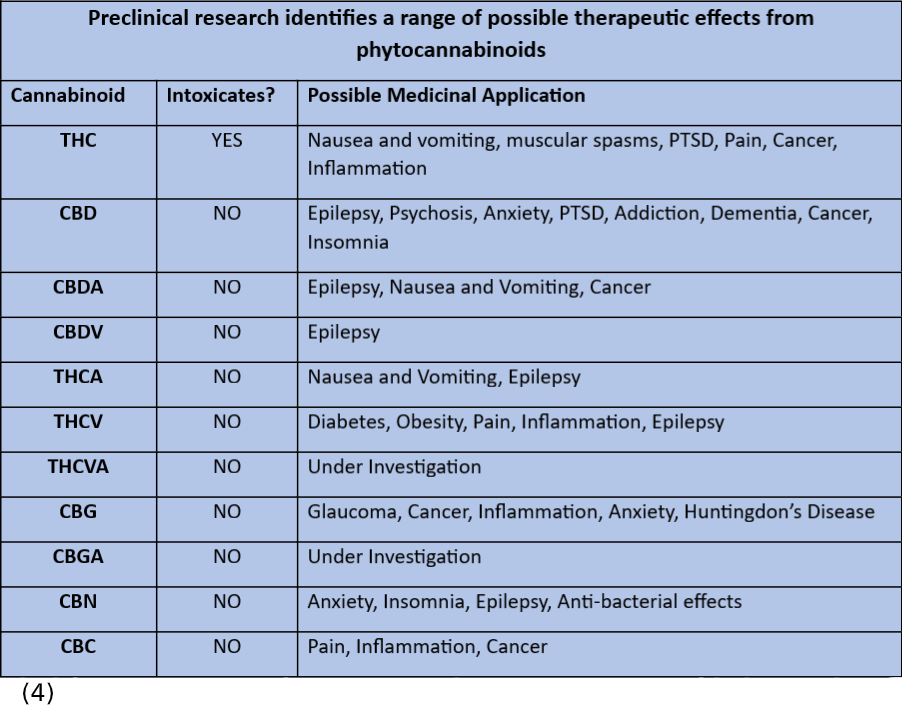
There is currently a lack of research evidence about the effectiveness of medical cannabis. For this reason the use of medical cannabis is only recommended when conventical treatments have been tried and proven unsuccessful in managing the person’s symptoms.
Multiple Sclerosis (MS)
Multiple sclerosis (MS) is a neurological condition. It affects the central nervous system (CMS – brain and spinal cord) causing a wide range of symptoms depending on where the CNS is affected. These symptoms can include problems with vision, arm or leg movement, sensation or balance. There are different types of MS that can sometimes cause serious disability, although it can occasionally be mild. In many cases, it's possible to manage symptoms and slow the progression of the disease.
Medical Cannabis has been used in the management of symptoms of MS. There is some quality research suggesting pharmaceutical grade THC and some THC:CBD ratios are effective in treating spasticity and pain in some patient groups. Research findings have been mixed about the effect of medical cannabis on improving urinary issues, sleep quality and disturbances and quality of life.
References:
Australian Government, Department of Health, Therapeutic Goods Administration (2017). Guidance for the use of medicinal cannabis in the treatment of multiple sclerosis in Australia. https://www.tga.gov.au/sites/default/files/guidance-use-medicinal-cannabis-treatment-multiple-sclerosis-australia.pdf viewed April 2020.
Medicinal Cannabis and MS. Ms Research Australia https://msra.org.au/medicinal-cannabis-ms/ viewed April 2020
Epilepsy
While up to 70 percent of people with epilepsy can obtain seizure control with medication, around about 30 percent continue to have seizures despite the availability of many antiepileptic drugs. This treatment gap has led people to seek alternatives, including medicinal cannabis. There is evidence that medical cannabis can be effective in epilepsy, especially for difficult to control seizures in syndromes such as Dravet Syndrome in children and Lennox-Gastaut Syndrome (LGS). Several studies have found low evidence for the use of medicinal cannabis products for the treatment of paediatric epilepsy, and for patients up to aged 25 years, especially when first-line treatments (anti-epileptic drugs) have been found to be ineffective.
The Therapeutic Goods Administration only recommends the treatment of epilepsy as an adjunct treatment in addition of existing anti-epilepsy medications.
Refer to C4E Cannabis for Epilepsy for further details. www.C4E.com.au
References:
Australian Government, Department of Health, Therapeutic Goods Administration (2017). Guidance for the use of medicinal cannabis in the treatment of epilepsy in paediatric and young adult patients in Australia.
https://www.tga.gov.au/sites/default/files/guidance-use-medicinal-cannabis-treatment-epilepsy-paediatric-and-young-adult-patients-australia.pdf viewed April 2020
Palliative care
Palliative care focuses on the quality of life of patients with life-threatening illness by providing relief from the symptoms, pain and stress. The use of medical cannabis products in palliative care is currently unclear. Cannabis as a palliative treatment for cancer patients seems to be well tolerated, effective and safe option to help patients cope with the malignancy related symptoms.
As there are very few studies on medicinal cannabis treatment in palliative care, it should be used only after standard treatments have failed. It is possible that medicinal cannabis will interact with chemotherapy and other medications used in palliative care.
Reference:
Australian Government, Department of Health, Therapeutic Goods Administration (2017). Guidance for the use of medicinal cannabis in the treatment of palliative care patients in Australia. https://www.tga.gov.au/sites/default/files/guidance-use-medicinal-cannabis-treatment-palliative-care-patients-australia.pdf viewed April 2020
Chemotherapy induced nausea and vomiting
A small number of studies have found relief of nausea and vomiting in patients with cancer who are undergoing chemotherapy.
While several studies found that the medicinal cannabis products were as effective as the prescription medicine it was compared with, most of the research studies were carried out some years ago, and in recent years much more effective prescription medicines for nausea and vomiting have become available. For these reasons, medical cannabis products for chemotherapy induced nausea and vomiting should be prescribed only after standard approved treatments have failed.
There are several clinical trials currently underway in Australia to investigate the effects of medical cannabis in specific areas of cancer care including loss of appetite, nausea, insomnia and pain.
References:
Cancer Council of Australia and Clinical Oncology Society of Australia: Medical use of cannabis position statement https://www.cancer.org.au/policy-and-advocacy/supportive-care-policy/cannabis-for-medical-purposes.html viewed April 2020
Australian Government, Department of Health, Therapeutic Goods Administration (2017). Guidance for the use of medical cannabis of the prevention or management of nausea and vomiting in Australia. https://www.tga.gov.au/sites/default/files/guidance-use-medicinal-cannabis-prevention-or-management-nausea-and-vomiting-australia.pdf viewed April 2020
Chronic non-cancer pain
There is moderate research confidence that patients with chronic non-cancer pain are likely to achieve and report reductions in pain rating compared with patients receiving a placebo.
The majority of the studies on the use of cannabinoids in pain have studied THC or THC-rich extracts. The studies are mainly where cannabinoids were used in addition to other pain medicines.
There is some evidence available for the treatment of neuropathic pain using medicinal cannabis products; however, the degree of effect is small. There is, however, insufficient information to make a conclusion about cannabinoids for the treatment of pain associated with arthritis and fibromyalgia.
There is much interest at present as to whether cannabinoids are "opioid sparing" – in other words, whether use of medicinal cannabis products for pain can result in a reduction of use of strong opioids. If this were the case, deaths and incapacity from opioid overdoses could be reduced, given that cannabinoids have fewer adverse outcomes. While some individuals with pain have reported that their use of opioids has been reduced when they also use medicinal cannabis, clinical studies in this area are still ongoing.
Reference:
Australian Government, Department of Health, Therapeutic Goods Administration (2017). Guidance for the use of medical cannabis in the treatment of chronic non-cancer pain. https://www.tga.gov.au/sites/default/files/guidance-use-medicinal-cannabis-treatment-chronic-non-cancer-pain-australia.pdf viewed April 2020